Sébastien Lecommandoux
Full professor
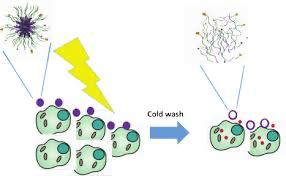
Description: Nanomedicine offers many advantages over classical forms of drug administration, the most important one being the targeting of high payloads of drugs to desired tissues. At the same time, this advantage entails a major drawback, namely the need to efficiently release active compounds from the carrier at the site of action. For current formulations this process is inefficient, limiting activity. Modalities for triggered release are urgently needed. Liposomes that decompose at increased temperature have been presented as one possible solution. For polymer-based nanoparticles no such solution exists in a clinical setting yet. The TEMPEAT project aims to overcome this limitation by developing targeted, thermo-responsive polypeptide particles for cancer treatment that combine a set of unique features: Temperature responsive: Nanocarriers based on stimulus-responsive elastin-like polypeptides (ELPs) are constructed by protein engineering. The ELPs reversibly assemble into stable nanoparticles at body temperature, but disassemble a few degrees below. Functionalized for targeting: ELPs are functionalized via protein engineering with nanobodies, selected for their efficient tumour targeting. By including nanobody-conjugated ELPs in the ELP polymer chain, these nanocarriers are made highly efficient at targeting tumours. Dual drug loading for efficacy: Two active compounds are loaded into the nanocarriers to potentiate cancer treatment. As one modality, photosensitizers are employed. Upon illumination, they induce short-lived oxygen radicals that kill cancer cells at micrometre distance, with high selectivity and specificity. In addition, anticancer agents are loaded into the particles’ core. Following targeted delivery to the cancer tissue, cooling leads to immediate release. We will focus on tumours in the peritoneal cavity, which constitute an enormous problem in metastasized ovarian cancer. This specific location allows in vivo cooling and laparoscopic illumination.
Collaborations: Jan van Hest (TUE, The Netherlands), Roland Brock and William Leenders (Radboud University, The Netherlands), Tomas Torres (Universidad Autonoma de Madrid, Spain), Catelijne Stortelers (Technology Ablynx, Belgium)