
Véronique Coma
Associate professor
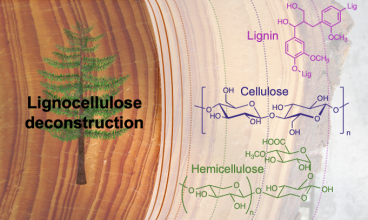
The increasing awareness concerning the environmental impact of fossil fuels leads to an increased interest in renewable resources. In this context, lignocellulose is foreseen as the most abundant renewable feedstock. Indeed, it contains the three biopolymers synthesized by biomass: cellulose, hemicelluloses and lignin. However, due to strong inter- and intra-molecular bonding, these polymers suffer from their very poor processability. Their fractionation and their conversion into high purity single components is mandatory to conceive their use in industrial processes. The resulting building block can then be used to synthesize biobased polymers with physico-chemical properties that match or exceed those of petroleum-based compounds.
In our team, we are actively contributing to this research field by developing new methods: (i) to ease the processability of lignocellulose-based polymers, (ii) to depolymerize those polymers, in a controlled fashion, in order to obtain valuable synthons, and (iii) to derivatize these synthons into functional monomers in order to develop new thermoplastic and thermosetting materials with desirable structure-property relationships.
Recent examples of our contribution to this field include the design of a CO2 switchable solvent system for the solubilization of cellulose in presence of a super base [1]. This new technic by-pass the use of non-renewable organic solvent such as dimethylformamide. The processability of cellulose was then performed through the chemical modification of its three hydroxyl groups, enabling the fabrication of transparent films with valuable mechanical properties [2]. 
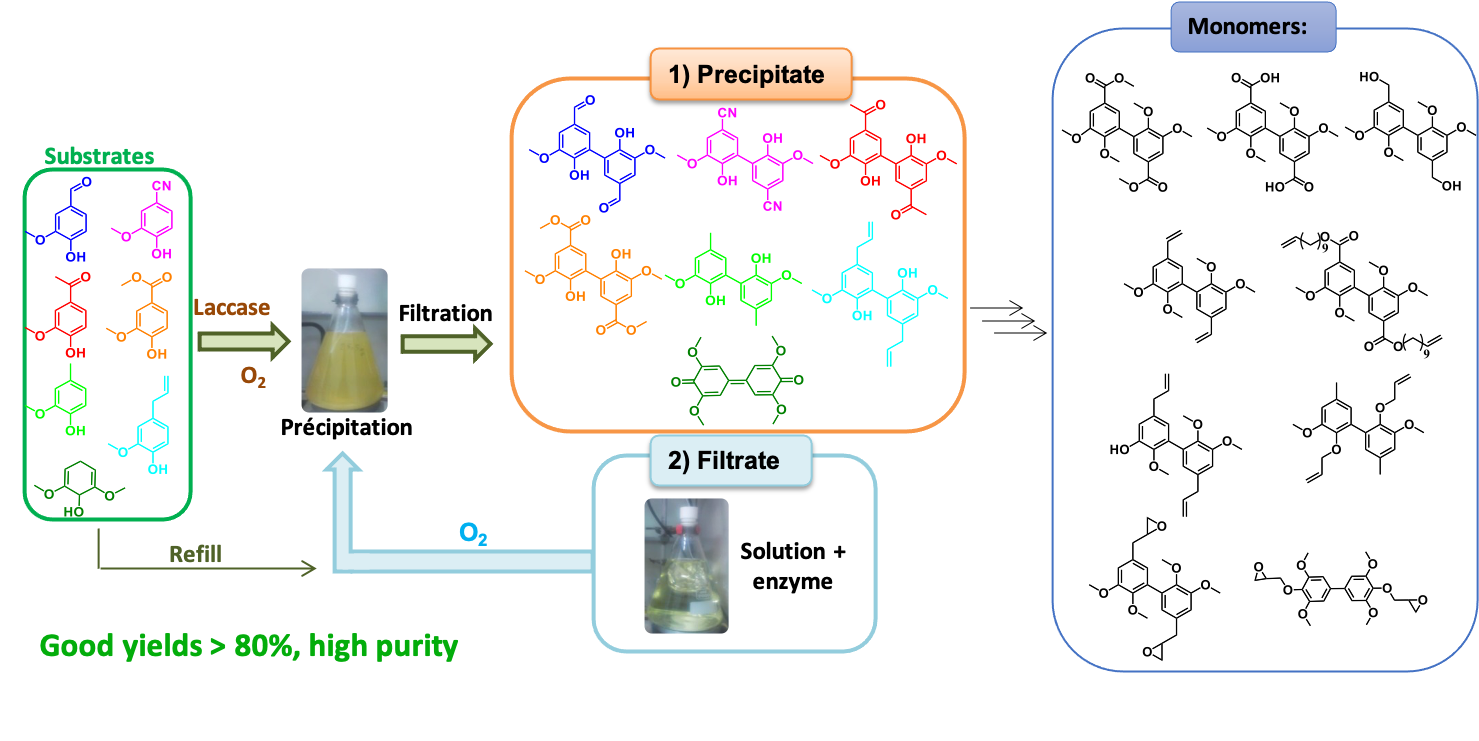
Another representative example is the laccase-catalyzed process to prepare, selectively, in high yield, dimers of lignin-based phenolic compounds without any purification [3]. These dimers were then used to synthesize different monomers such as polyols, polyepoxyde, polyacide, that are envisioned as substitutes of Bis-phenol-A. This is an industrially-relevant method to valorize synthons obtained from lignin depolymerization, notably vanillin.
[1] K.N. Onwukamike, T. Tassaing, S. Grelier, E. Grau, H. Cramail, M.A.R. Meier, ACS Sustain. Chem. Eng., 6(1), 1496-1503 (2017)
[2] K.N. Onwukamike, S. Grelier, E. Grau, H. Cramail, M.A.R. Meier, ACS Sustain. Chem. Eng., 6, 8826-8835 (2018)
[3] A. Llevot, E. Grau, S. Carlotti, S. Grelier, H. Cramail, J. Mol. Catal. B-Enzym., 125, 34-41 (2016)